Understanding a cerium quirk could help advance grid-scale energy storage
It turns out cerium flow batteries lose voltage when electrolyte molecules siphon off energy to form different complexes around the metal.

It turns out cerium flow batteries lose voltage when electrolyte molecules siphon off energy to form different complexes around the metal.
An explanation for why flow batteries using the metal cerium in a sulfuric acid electrolyte fall short on voltage, discovered through a study led by the University of Michigan, could pave the way for better battery chemistry.
Flow batteries are one of the methods under consideration for storing intermittent sources of renewable electricity, such as solar and wind power. They can bank large quantities of energy by keeping the chemical potential in liquid form, with two electrolytes that flow through porous electrodes to charge and discharge. The metal cerium could store energy at a relatively high voltage, meaning more energy per metal ion, and at low cost.
One of the challenges with cerium is figuring out how to make electric charges transfer to and from the electrode efficiently. On its way through the positive electrode, cerium either picks up or drops off an electron, depending on whether the battery is charging or discharging.

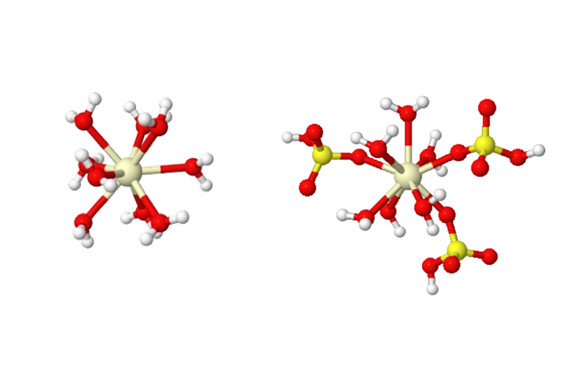
However, the cerium in a sulfuric acid electrolyte doesn’t pick up and drop off the electron as quickly as expected, meaning energy is wasted. It turned out that the water molecules and sulfate molecules were doing a complicated dance around the cerium, and that’s how the energy was lost.
“Through this study, we have a better understanding of how cerium ions behave in acidic electrolytes during charge transfer,” said Cailin Buchanan, a recent U-M doctoral graduate in chemical engineering and first author of the paper in the journal JACS Au.
“This understanding will help us and future researchers to design more efficient cerium-based batteries that have less voltage loss during charging and discharging.”
The team took a close look at what was happening when cerium picked up and dropped off electrons, using X-ray absorption to keep an eye on bonds and associations between the cerium, sulfates and water. These experiments were done at Argonne National Laboratory. They followed those measurements with computer simulations, led by Bryan Goldsmith, the Dow Corning Assistant Professor of Chemical Engineering at U-M.
“We find that when cerium is short three electrons, it is only surrounded by water molecules, while when it gives up that fourth electron, sulfate or bisulfate ions are hanging off the cerium ion,” said Nirala Singh, U-M assistant professor of chemical engineering and corresponding author of the study, who led the experiment.
“As a result of this, when we oxidize cerium by taking away that electron, or reduce it by giving the electron back, both an electron transfer has to occur and the molecules around it have to rearrange.”
By understanding the energy associated with this structural rearrangement, the researchers were able to explain why the reaction is asymmetrical, where the oxidation and reduction behave differently. Because of this, the go-to theory for predicting the rates of electron transfer, known as Marcus theory, isn’t enough. Instead, the team found that it’s possible to use Marcus theory to figure out the electron transfer piece and then add in the effects of rearrangement in a two-step process.
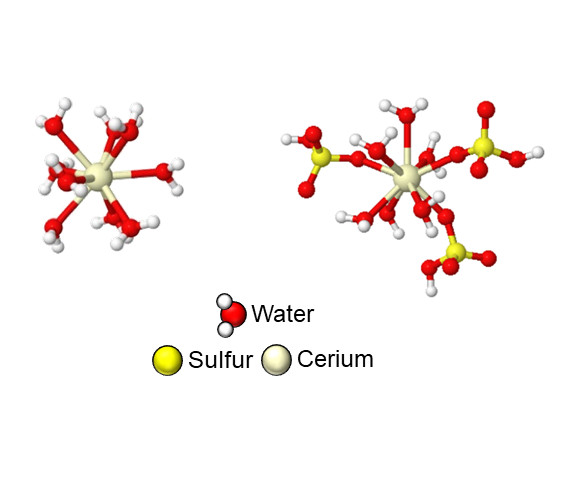
“The uneven complexation between the oxidized and reduced forms of cerium causes reaction rates to slow down, and this knowledge will inform electrolyte design strategies for cerium or other similar flow batteries,” Singh said.
Using the team’s two-step method, researchers will be able to identify electrolytes that have fast reaction rates, yielding high efficiencies. Ultimately, the goal is to use electrolytes that won’t store different amounts of energy in the complexes around the oxidized or reduced cerium ion.
In addition to opening new avenues in grid-scale energy storage, this discovery could improve other chemical processes that rely on cerium, such as the manufacturing of carbon-based products and wastewater decontamination.
The research is supported by the University of Michigan and the Dow Sustainability Fellows Program. Electrodes to prepare the cerium ions were provided by De Nora.
Computing resources were provided by the Department of Energy’s National Energy Research Scientific Computing Center and the National Science Foundation’s Extreme Science and Engineering Discovery Environment.