
$2.38M to test nano-engineered brain cancer treatment in mice
A protein that crosses the blood-brain barrier carries a drug that kills tumor cells and another that activates the immune system.

A protein that crosses the blood-brain barrier carries a drug that kills tumor cells and another that activates the immune system.
U-M EXPERTS:
Originally written by Kate McAlpine and published by Michigan Engineering.
A new nanomedicine that crosses the blood-brain barrier, engages the immune system and kills cancer cells may offer hope for treating the most aggressive form of brain cancer, glioblastoma. With $2.38 million in funding from the National Institutes of Health, the medicine will soon be tested in mice at the University of Michigan.
Led by a nano-engineer and neuro-oncology researchers at U-M, the study is the first to test the two drugs together, packaged so that they can be delivered through the bloodstream rather than a hole in the skull. It builds on previous success eliminating cancer in seven out of eight mice by packaging just the immune drug in the protein that crosses the blood-brain barrier so that it could be delivered intravenously. The 5-year survival rate for glioblastoma in humans is about 5%.
“The standard of care for glioblastoma is surgery and radiation, and the median survival hasn’t improved for several decades. A systemically delivered nanomedicine that can prolong survival and prevent recurrence is the dream,” said Maria Castro, the R. C. Schneider Collegiate Professor and a professor of neurosurgery. Her team leads the mouse studies in collaboration with Pedro Lowenstein, the Richard Schneider Collegiate Professor and a professor of neurosurgery.
As the team tests out the nanoparticles timed to release the immune drug followed by a tumor-killing drug, developed and produced by Lahann’s group, one of the key questions is how well the drugs cooperate.
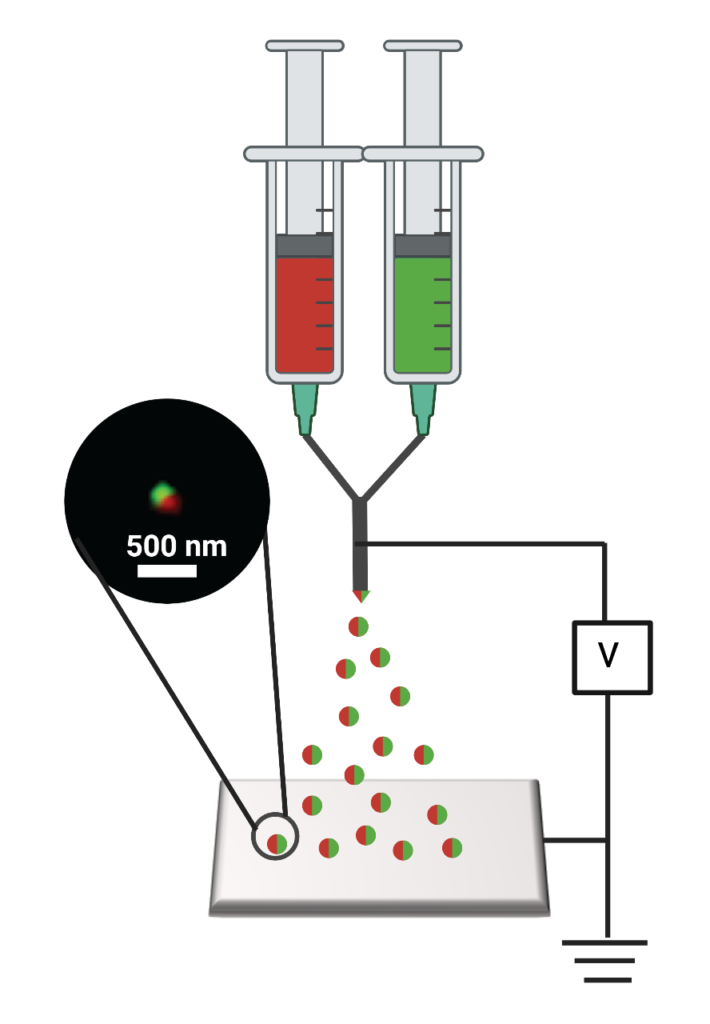
“Are they working much better than either drug alone? That’s what we’re hoping for. Or is it just a small improvement—or are they actually competing with each other and making the treatment worse or increasing the side effects?” said Joerg Lahann, the Wolfgang Pauli Collegiate Professor of Engineering and project lead.
The advanced nanomedicines are delivered intravenously and combined with radiation therapy, as they would be in a future clinical trial.
To get the nanomedicine from the bloodstream to the brain, Lahann’s team packages the drugs in a protein called human serum albumin, which is present in blood and can cross the blood-brain barrier. Once there, the drugs must wake up the immune system to prevent recurrence and death, which frequently follow conventional treatments like surgery, radiation and chemotherapy.
Tumors grow and regrow because cancer cells have ways of suppressing the immune system. The 2020 study and the new grant use a drug that blocks STAT3, a signaling molecule that cancer cells use to tell immune cells not to attack them. This gave the immune system of the mice the ability to identify the cancer cells as targets for destruction.
In a study just out in May, the team used a drug that blocks CXCR4, an immune receptor that receives orders to send killer T-cells away. Blocking CXCR4 helps keep T-cells in the brain, where they do their work of killing brain cancer cells. Three out of five mice survived long-term, and all of those survivors cleared new tumors during the recurrence challenge. While the new grant won’t use this drug, the team is interested in a future study exploring whether two immune approaches together might be more effective.
“Tumors have a lot of variation, so we need to attack them from many directions,” said Lowenstein.
After initial testing of the new two-compartment nanomedicine in lab-grown cell cultures that mimic human tumors and their surroundings, the team will begin testing in mice as the next step toward clinical trials in humans. They will find out how much of the nanomedicine makes it into the brain, how well it fights the cancer, how well it leaves the body and what the side effects are like. The previous studies suggest that the nanoparticles home in on tumor cells, infiltrating them more often than healthy cells, and one of the goals for this one is to better understand how that works.
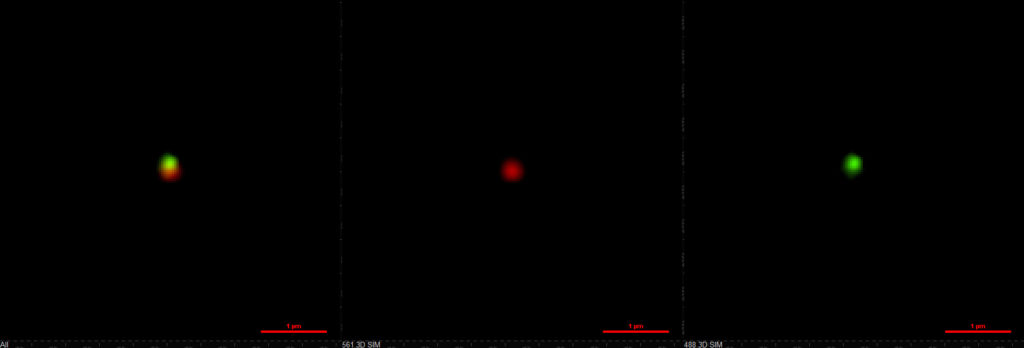
“For nanomedicines to advance into clinical trials as experimental treatments for glioblastoma, we must understand the mechanisms by which they accumulate in tumor and other tissues,” said Colin Greineder, an assistant professor of emergency medicine at U-M, who will lead studies of how the nanomedicine distributes in the body.
Lahann is also the director of the Biointerfaces Institute at U-M. Castro and Lowenstein are also professors of cell and developmental biology.